Four Corners also uncovered under-reporting of adverse impacts of stimulators in the TGA's database, which Health Minister Mr Butler described as "shocking" claims.Īustralian sponsors of medical devices are legally required to report adverse events to the TGA. "That then becomes a big market potentially for pharmaceutical companies and implanted device companies," he says."I found that people were just not making good improvements," he says. Orthopaedic surgeon Professor Ian Harris co-authored a research paper on them last year and says they lack credible scientific evidence as a treatment for chronic back pain."They're probably not better than a placebo procedure … we don't have good evidence on the rate of complications or just how dangerous they are. The TGA launched a post-market review into spinal cord stimulators in 2022, but the results are yet to be published. Over the next few years, Gordon had revision surgery to fix complications with the device and had one replaced. "You'll lose control of your proper bowel functions … bladder function, you'll lose sexual function. 
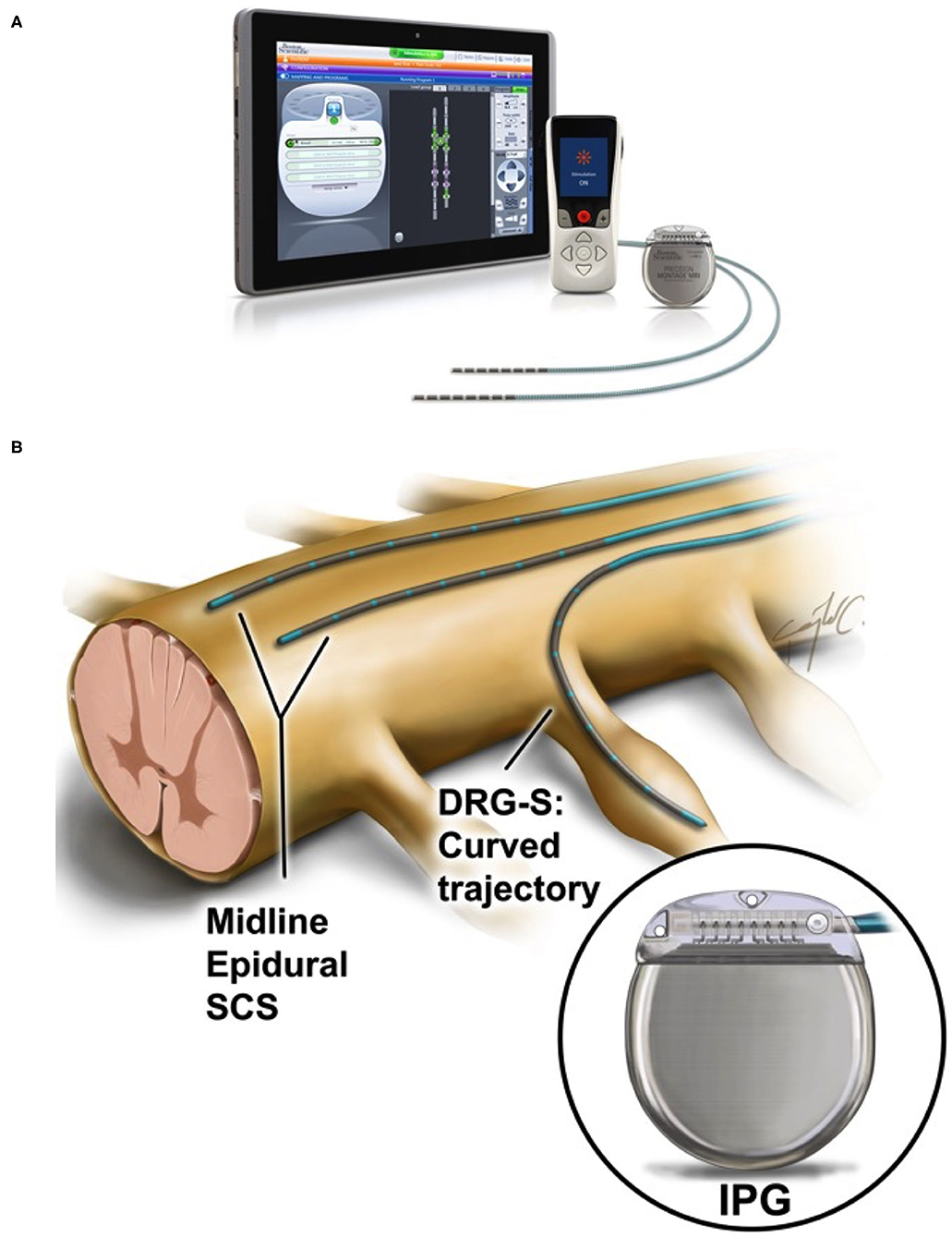
That's me being able to go running again,'" Teresa says.A spinal cord stimulator, or "SCS", is a small medical device that can be recommended for patients with back pain. "I heard that and I thought, 'my god, that's life-changing. A Four Corners investigation has exposed the harms spinal cord stimulator s, a treatment for back pain, have caused some people 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
